Did you know cephalopods may have traded evolution gains for extra smarts? I didn’t either. I don’t believe it, anyway. The paper is fine, though, it’s just the weird spin the media has been putting on it.
The actual title of the paper is Trade-off between Transcriptome Plasticity and Genome Evolution in Cephalopods, which is a lot more accurate. The authors discovered that there’s a lot of RNA editing going on in coleoids. The process is not a surprise, we’ve known about RNA editing for a long time, but the extent in squid is unusual.
RNA editing is basic college-level stuff, so if your experience in biology is limited to high school classes or pop press summaries, you may not be familiar with it, so a quick summary follows.
There is a small family of enzymes in vertebrates called the ADARs, or Adenosine Deaminases Acting on RNA. These enzymes bind to double-stranded RNA, and convert the adenosine bases to inosine. Inosine preferentially base-pairs with cytosine, so this functionally converts the As in the double-stranded stretch into Gs.
There are limitations on this enzyme, so it doesn’t charge off and convert every A in every RNA into a G, which would be lethal! Since it only works on double-stranded RNA, which requires that the sequence of the RNA be such that it can fold back on itself and create regions where long bits of the sequence are binding to itself, it only works on some RNAs. This requirement also means that in some ways it is evolutionarily and genetically fragile — mutate a base somewhere in that dsRNA stretch, and it stops being double-stranded, and the enzyme no longer converts A to G.
Before the creationists and ID creationists get all excited, this is not revolutionary, and does not in some way preclude evolution. The presence of these enzymes means that there is an alternative way to tweak some bases in a sequence other than by directly modifying it by mutation in the genome. In a simplistic way, you can just think of it as another way a single-base pair mutation can occur — it’s just done in the transcriptome, rather than the genome.
It also has some advantages. Because the enzyme is not perfect, some RNAs escape the modification, which means the organism can have both the unedited and edited forms of the protein — so if the unedited form performs some useful function, that function hasn’t been eliminated in one swoop. It may provide a kind of soft transition between two forms of a protein.
There is also a disadvantage. Because the formation of double-stranded RNA requires the maintenance and cooperation of multiple bases in multiple regions of the sequence, coming to rely on RNA editing for one base can lock in the sequence of multiple other bases. That’s the meaning of the title of the article: the tradeoff is that yes, you can get a useful adaptive modification by modifying the transcriptome (transcriptome plasticity), but if you’re dependent on that, you’ve also limited the amount of change you can tolerate in associated regions of the genome (genome evolution). There is basically a window around the change you want that is approximately 200 bases long that you now have to constrain in order for your key change to continue to work.
That’s limiting. The authors point out that only about 3% of human RNA messages are recoded by ADARs, and only about 25 total are conserved across mammals. That implies that a lot of the RNA editing going on is spurious, but some tiny part of it is functionally necessary.
In contrast, some cephalopods, the squid and octopuses, have many more.
The authors searched for non-synonymous sequences between RNA and DNA. They found lots, and further, the A-to-G mismatches, which is what you’d expect if this were a result of ADAR editing, were greatly enriched. Most importantly, they compared multiple species, and only the coleoids exhibited this pattern, while Nautilus and Aplysia did not. This is a property of the organisms, not an artifact of their methods.
So how many sequences are modified by ADARs in squid? That’s tough to determine, since we know there will be spurious editing all over the place, but we can say confidently that it’s orders of magnitude more than what is seen in humans. The most relevant and conservative estimate in the paper is that “1,146 editing sites (in 443 proteins) are conserved and shared by all four coleoid cephalopod species”. Compare that to the 25 in mammals.
Also interesting is that the sequences identified are often developmentally and neurobiologically significant. Protocadherins (molecules involved in cell adhesion, and therefore important in the development of multicellular animals) were enriched both in overall number and in RNA editing sites. They also specifically compared the function of neuronal channel proteins in both the edited and the unedited versions. The unedited proteins were still functional, but the edited versions were more sharply tuned in their electrical properties.
Here’s an example. They recorded the potassium current for unedited K+ channels on the left, and for edited channels on the right. These are conserved editing sequences maintained in squid, cuttlefish, and octopus, and you can clearly see that editing removes differences between those species to produce a more similar pattern of current flow.
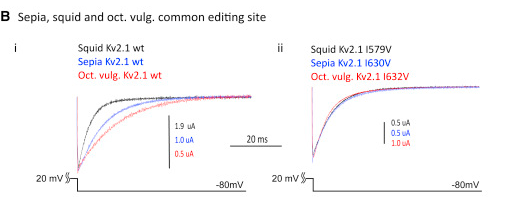
Conserved and Species-Specific Editing Sites Affect Protein Function
Unedited (WT) and singly edited versions of the voltage-dependent K+ channels of the Kv2 subfamily were studied under voltage clamp.
(B) (i) Tail currents measured at a voltage (Vm) of −80 mV, following an activating pulse of +20 mV for 25 ms. Traces are shown for the WT Kv2.1 channels from squid, sepia, and Octopus vulgaris. (ii) Tail currents for the same channels edited at the shared I-to-V site in the 6th transmembrane span, following the same voltage protocol.
That we’re seeing changes in nervous system activity is probably why the first article I cited seems to think this has something to do with making cephalopods smarter. It doesn’t, directly. It’s more that one of the mechanisms driving the cephalopod radiation after the Cambrian was adoption of modification of the transcriptome via RNA editing. It’s a pattern that isn’t easily reversed — a lot of proteins would have to be tweaked at the genome level to make them independent of ADARs — but it works, so there’s no particular pressure on them to modify the mechanism.
Liscovitch-Brauer, Noa et al. (2017) Trade-off between Transcriptome Plasticity and Genome Evolution in Cephalopods. Cell 169(2):191-202.


Not all the cephalopods — it looks like they found it only in coleoids, and explicitly NOT in Nautilus because they looked. No Spirula though, alas. Maybe someone else can check, or maybe the authors will. That’d be nice
Either Nautilus has lost it (possible!), in which case it could be as old as stem-group cephs; or maybe it’s confined to the coleoids, which would pull it quite a bit later than the Cambrian — that would put it probably in the Mississippian, or even later if the belemnites didn’t have it. (Cephalopod taxonomy on the class-to-order level is kind of a mess, unfortunately.) My question is, as always, what about ammonoids???
For the Moron in Chief, this will justify a certain Stargate program.
For the victims, likely, zero rights.
Expand upon that, as is “necessary for national security”, to ensure silencing of any research on genetics.
I’m far from paranoid, but do have some degree of experience with National Security Programs.
Christ, would that he actually existed, we all need help! Considering the program levels and destruction of records to avoid oversight.
I’ll not at all say anything over a third reason. We have no desire to join Manning.
wzrd1@2,
RNA editing is a matter of national security?
Spirula is a pretty ordinary coleoid.
I’ll also mention that if you read the paragraph starting “The authors searched for non-synonymous sequences…”, I specifically pointed out that they didn’t see this expansion of edited sequences in non-coleoid cephalopods and Aplysia.
It’s kind of weird to expect them to work on Spirula. They examined Octopus vulgaris, Octopus bimaculoides, Doryteuthis pealeii, Sepia oficianalis,Nautilus pompilius, Aplysia californica — can’t expect them to do every cephalopod species.
If there’s an omission, it’s Euprymna. That’s a common genus that’s seeing a lot of laboratory study.
I should say that #2 was a joke, for those uncertain.
I also entered the post on the wrong tab. That’s what I get for having multiple tabs open and taking my glasses off. :/
As for RNA editing, I find it fascinating that “broken” DNA, which then makes defective RNA, which makes defective proteins gets the protein recoded into an operational configuration. That is one astounding adaptation! Instead of intelligent design, we get a natural design that is intelligent in its own right, biochemically speaking.
I never fail to be amazed what can be done with a mere four “letters”!
Huh, what an interesting result. I wonder what they would find if they tested Micropilina.